Multiscale Modeling of Pulmonary Arterial Hypertension
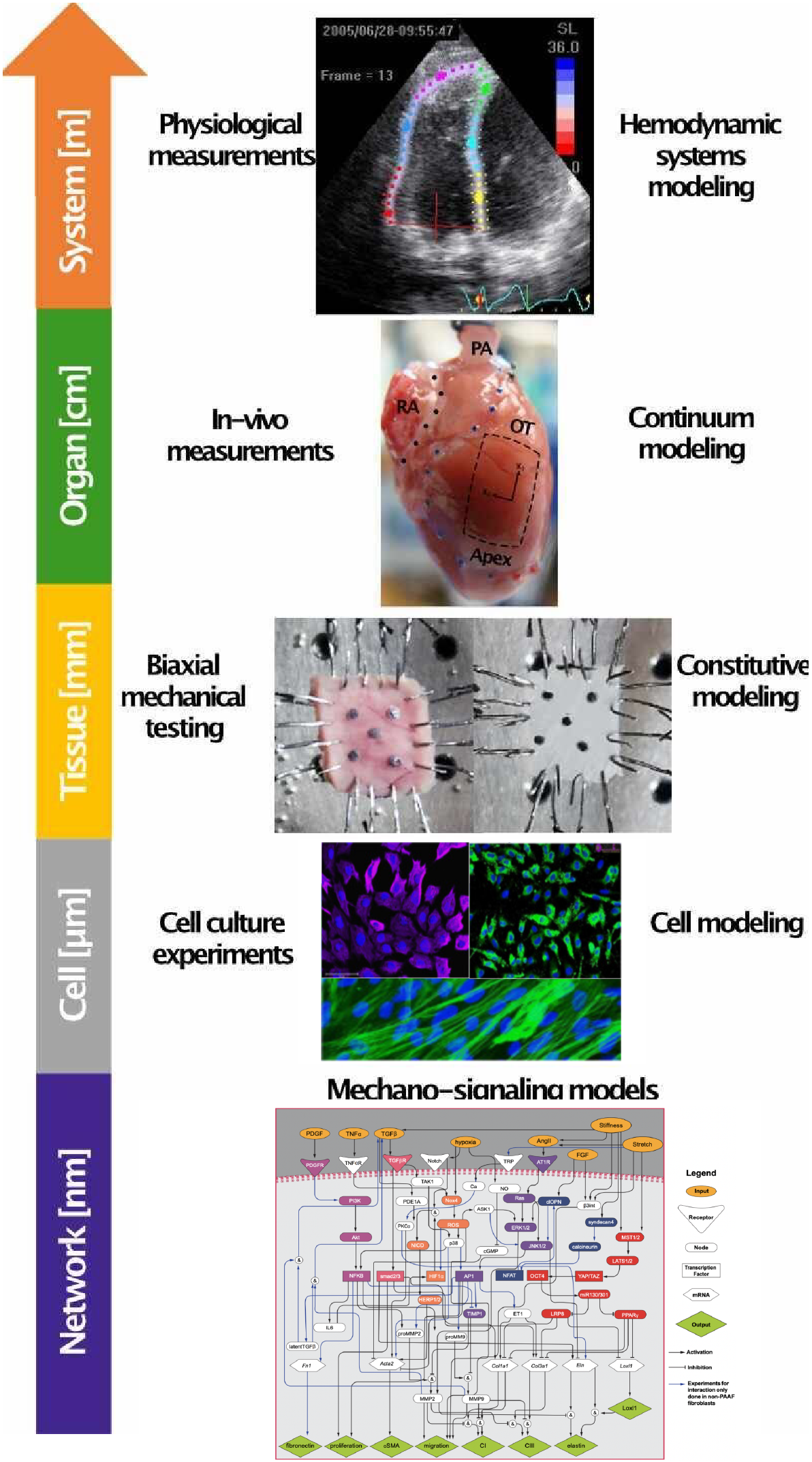
Pulmonary Arterial Hypertension
Sex matters: Pulmonary arterial hypertension (PAH) is caused by adverse idiopathic remodeling of the pulmonary arteries, is ~3X more prevalent in women, and results in 5-year survival <50% due to right-ventricular (RV) failure. There are no therapies to prevent RV failure or reverse vascular remodeling in PAH. In the DVJ Lab, we study male and female rats, and investigate the role of ovarian hormones by including ovariectomized female animals.

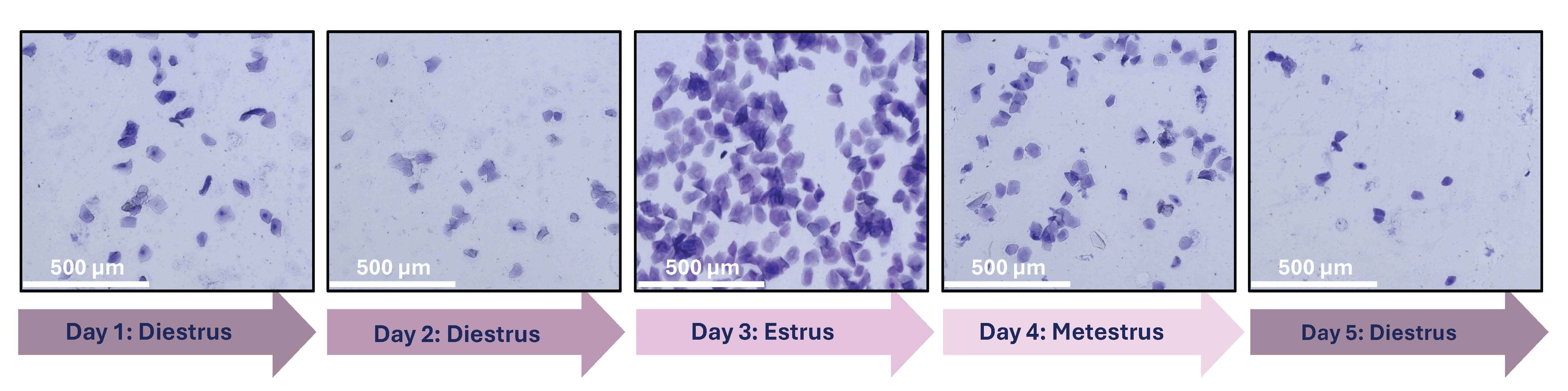
Accounting for the Estrous Cycle in Our Studies
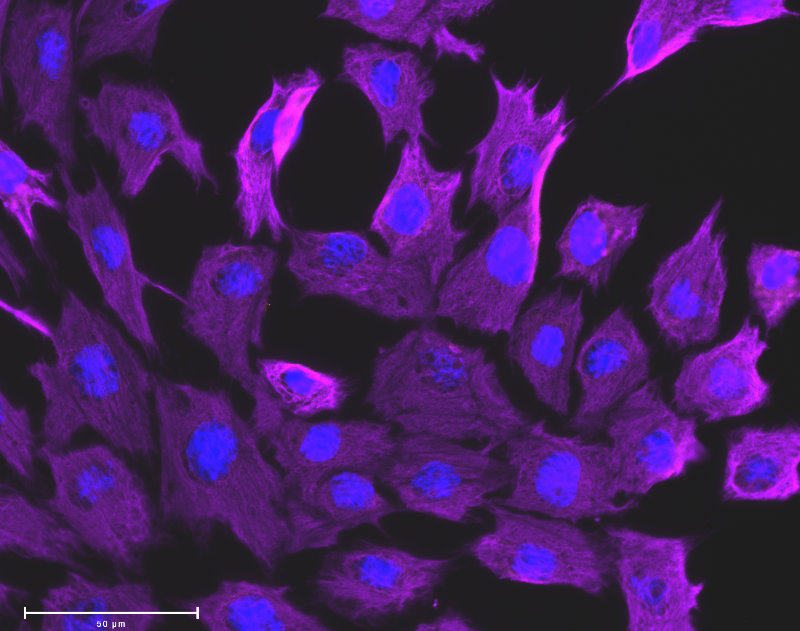
Mechanobiology
We have shown that mechanical stimuli regulate the phenotypes of the cells involved in cardiac and vascular remodeling in PAH. In particular, the roles of stretch and altered ECM stiffness in regulating vascular and cardiac fibrosis are being investigated. Cell signaling data are synthesized into network models that we use to investigate the systems biology of mechanoregulatory pathways in the pulmonary arteries and myocardium in PAH.

Right-Ventricular Mechanics
Here, we use a planar biaxial testing protocol to obtain stress-strain measurements of intact (fully cellularized) and decellularized RV samples. By measuring structural changes in the tissues during PAH, we build microstructural constitutive models and elucidate structure-function relations during the progression of PAH.
Vélez-Rendón et al. Journal of Biomechanical Engineering 2019

Vascular Mechanics
We are interested in modeling the mechanical properties of the left and right pulmonary arteries in normo- and hypertensive animal models. The harvested vessels are subjected to tubular biaxial testing in the axial and circumferential directions. The resulting dynamic stress-strain relation is analyzed and used to model the vascular adaptations and hemodynamic consequences of disease progression of PAH.

Extracellular Matrix Properties
We have shown that extracellular matrix (ECM) remodeling of the pulmonary arteries and right ventricle is important in the pathogenesis of PAH. We use quantitative microscopy, biaxial tissue of intact and decellularized tissues, and microstructural constitutive models to investigate how changes in tissue structure affect the structure and biomechanics of the vessels and myocardium. We also use in vitro studies to investigate the cellular mechanisms of tissue fibrosis.

Hemodynamic Studies
We carry out in vivo hemodynamic measurements to study right-ventricular and pulmonary vascular physiology and their biomechanical interactions in an animal model of pulmonary arterial hypertension (PAH). These hemodynamic data inform computational models of the pathophysiology of PAH.

Wu Tsai Human Performance Alliance at UC San Diego
Project 4 Biomechanics
To study the mechanisms by which elite athletes achieve optimal musculoskeletal performance, we are measuring the mechanical properties of musculoskeletal tissues from animal models of exercise and human subjects so that we can develop mechanistic multiscale models in the Multiscale moonshot of the Wu Tsai Human Performance Alliance